VIVO Pathophysiology
Other Topics
Osmosis, Tonicity, and Hydrostatic Pressure
Large quantities of water molecules constantly move across cell membranes by simple diffusion, often facilitated by movement through membrane proteins, including aquaporins. In general, net movement of water into or out of cells is negligible. For example, it has been estimated that an amount of water equivalent to roughly 100 times the volume of the cell diffuses across the red blood cell membrane every second; the cell doesn't lose or gain water because equal amounts go in and out.
There are, however, many cases in which net flow of water occurs across cell membranes and sheets of cells. An example of great importance to you is the secretion of and absorption of water in your small intestine. In such situations, water still moves across membranes by simple diffusion, but the process is important enough to warrant a distinct name - osmosis.
Osmosis and Net Movement of Water
Osmosis is the net movement of water across a selectively permeable membrane driven by a difference in solute concentrations on the two sides of the membrane. A selectively permiable membrane is one that allows unrestricted passage of water, but not solute molecules or ions.
Different concentrations of solute molecules leads to different concentrations of free water molecules on either side of the membrane. On the side of the membrane with higher free water concentration (i.e. a lower concentration of solute), more water molecules will strike the pores in the membrane in a give interval of time. More strikes equates to more molecules passing through the pores, which in turn results in net diffusion of water from the compartment with high concentration of free water to that with low concentration of free water.
The key to remember about osmosis is that water flows from the solution with the lower solute concentration into the solution with higher solute concentration. This means that water flows in response to differences in molarity across a membrane. The size of the solute particles does not influence osmosis. Equilibrium is reached once sufficient water has moved to equalize the solute concentration on both sides of the membrane, and at that point, net flow of water ceases. Here is a simple example to illustrate these principles:
| Two containers of equal volume are separated by a membrane that allows free passage of water, but totally restricts passage of solute molecules. Solution A has 3 molecules of the protein albumin (molecular weight 66,000) and Solution B contains 15 molecules of glucose (molecular weight 180). Into which compartment will water flow, or will there be no net movement of water? [ answer ] | 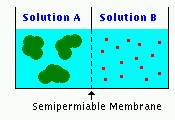 |
Additional examples are provided on how to determine which direction water will flow in different circumstances.
Tonicity
When thinking about osmosis, we are always comparing solute concentrations between two solutions, and some standard terminology is commonly used to describe these differences:
- Isotonic: The solutions being compared have equal concentration of solutes.
- Hypertonic: The solution with the higher concentration of solutes.
- Hypotonic: The solution with the lower concentration of solutes.

In the examples above, Solutions A and B are isotonic (with each other), Solutions A and B are both hypertonic compared to Solution C, and Solution C is hypotonic relative to Solutions A and B.
Diffusion of water across a membrane generates a pressure called osmotic pressure. If the pressure in the compartment into which water is flowing is raised to the equivalent of the osmotic pressure, movement of water will stop. This pressure is often called hydrostatic ('water-stopping') pressure. The term osmolarity is used to describe the number of solute particles in a volume of fluid. Osmoles are used to describe the concentration in terms of number of particles - a 1 osmolar solution contains 1 mole of osmotically-active particles (molecules and ions) per liter.
The classic demonstration of osmosis and osmotic pressure is to immerse red blood cells in solutions of varying osmolarity and watch what happens. Blood serum is isotonic with respect to the cytoplasm, and red cells in that solution assume the shape of a biconcave disk. To prepare the images shown below, red cells from your intrepid author were suspended in three types of solutions:
- Isotonic - the cells were diluted in serum: Note the beautiful biconcave shape of the cells as they circulate in blood.
- Hypotonic - the cells in serum were diluted in water: At 200 milliosmols (mOs), the cells are visibly swollen and have lost their biconcave shape, and at 100 mOs, most have swollen so much that they have ruptured, leaving what are called red blood cell ghosts. In a hypotonic solution, water rushes into cells.
- Hypertonic - A concentrated solution of NaCl was mixed with the cells and serum to increase osmolarity: At 400 mOs and especially at 500 mOs, water has flowed out of the cells, causing them to collapse and assume the spiky appearance you see.


Predict what would happen if you mixed sufficient water with the 500 mOs sample shown above to reduce its osmolarity to about 300 mOs.
Calculating Osmotic and Hydrostatic Pressure
The flow of water across a membrane in response to differing concentrations of solutes on either side - osmosis - generates a pressure across the membrane called osmotic pressure. Osmotic pressure is defined as the hydrostatic pressure required to stop the flow of water, and thus, osmotic and hydrostatic pressures are, for all intents and purposes, equivalent. The membrane being referred to here can be an artifical lipid bilayer, a plasma membrane or a layer of cells.
The osmotic pressure P of a dilute solution is approximated by the following:
where R is the gas constant (0.082 liter-atmosphere/degree-mole), T is the absolute temperature, and C1 ... Cn are the molar concentrations of all solutes (ions and molecules).
Similarly, the osmotic pressure across of membrane separating two solutions is:
where ΔC is the difference in solute concentration between the two solutions. Thus, if the membrane is permeable to water and not solutes, osmotic pressure is proportional to the difference in solute concentration across the membrane (the proportionality factor is RT).
Advanced and Supplemental Topics
Send comments to Richard.Bowen@colostate.edu