VIVO Pathophysiology
Extracellular Calcium-Sensing Receptor
Maintaining tight control over the concentration of calcium in blood and extracellular fluid is a critical task. It stands to reason that a calcium sensor would evolve as a component of the system responsible for calcium homeostasis. Considering its involvement in modulating so many physiologic processes, calcium itself can be thought of as a type of hormone, and the calcium sensor as its receptor.
The DNA sequence encoding the extracellular calcium sensor was originally isolated from bovine parathyroid gland. Since then, corresponding sequences have been isolated from a broad range of species, enabling serious study of this intriguing membrane protein.
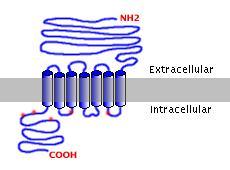
The calcium-sensing receptor is a member of the G protein-coupled receptor family. Like other family members, it contains seven hydrophobic helices that anchor it in the plasma membrane. The large (~600 amino acids) extracellular domain is known to be critical to interactions with extracellular calcium. The receptor also has a rather large (~200 amino acids) cytosolic tail. These features are depicted in the figure to the right; the red highlights on the intracellular domain correspond to potential protein kinase phosphorylation sites.
Activation of the calcium sensor has two major signal-transducing effects:
- Activation of phospholipase C, which leads to generation of the second messengers diacylglycerol and inositol trisphosphate.
- Inhibition of adenylate cyclase, which suppresses intracellular concentration of cyclic AMP.
The sensor can also activate the mitogen-activated protein kinase pathway, suggesting an ability to influence nuclear function.
The calcium sensor is expressed in a broad range of cells, including parathyroid cells and C cells in the thyroid gland, indicating its involvement in controlling the synthesis and secretion of parathyroid hormone and calcitonin. Functional studies and investigation of animals with mutations in the calcium sensor gene have confirmed that the calcium sensor directly affects secretion of these two hormones.
The calcium sensor is also expressed in several cell types in the kidney, osteoblasts, a variety of hematopoietic cells in bone marrow, and in the gastrointestinal mucosa. Strangely, it is also present in the squamous epithelial cells of the esophagus. Such a broad distribution of expression supports that concept that calcium, acting as a hormone, has direct effects on the function of many cell types.
Understanding the role of the calcium sensor in calcium homeostasis has benefited greatly by the study of mutations in the human gene encoding this receptor:
- Inactivating mutations: A large number of different mutations in the calcium sensor gene have been identified in patients affected by a type of calcium resistance known as familial hypocalciuric hypercalcemia. The different mutations result in a spectrum of calcium sensor dynfunctions, ranging from total inactivation to a moderate decrease in affinity of the receptor for calcium. A prominent clinical consequence of such mutations is an abnormal set point or sensitivity of the parathyroid gland to blood calcium concentration. Abnormalities in renal excretion of calcium are also observed.
- Activating mutations: Certain types of mutations lead to a calcium sensor with an elevated sensitivity to calcium. The clinical consequence of such mutations is familial hypercalciuric hypocalcemia - basically the opposite of what is seen with inactivating mutations in the sensor gene. High blood calcium affects the normal calcium sensor to suppress parathyroid hormone secretion. If the calcium sensor is constitutively more active than, a type of hypoparathyroidism results.
Further study of the extracellular calcium-sensing receptor will undoubtedly shed further light on the intricacies of how calcium homeostasis is regulated, and may provide new opportunities for treatment of diseases of calcium metabolism.
References and Reviews
- Brown EM: Physiology and pathophysiology of the extracellular calcium-sensing receptor. Amer J Med 106:238-253, 1999.
- Brown EM, Gamba G, Riccardi D, etc: Cloning and characterization of an extracellular Ca2+-sensing receptor from bovine parathyroid. Nature 366:575, 1993.
Send comments to Richard.Bowen@colostate.edu
