VIVO Pathophysiology
Mineralocorticoids
Removal of the adrenal glands can lead to death within just a few days. Observation of such a unfortunate subject would reveal several key derangements:
- the concentration of potassium in extracelluar fluid becomes dramatically elevated
- urinary excretion of sodium is high and the concentration of sodium in extracellular fluid decreases significantly
- volume of extracellular fluid and blood decrease
- the heart begins to function poorly, cardiac output declines and shock ensues
These phenomena are a direct result of loss of mineralocorticoid activity, and can largely be prevented by replacement of salts and mineralocorticoids. Clearly mineralocorticoids are acutely critical for maintenance of life!
Aldosterone and Mineralocorticoid Receptors
The principal steroid with mineralocorticoid activity is aldosterone. Cortisol, the major glucocorticoid in non-rodent species, is said to have "weak mineralocorticoid activity", which is of some importance because cortisol is secreted very much more abundantly than aldosterone. Another way to state this is that a small fraction of the mineralocorticoid response in the body is due to cortisol rather than aldosterone.
The mineralocorticoid receptor binds both aldosterone and cortisol with equal affinity. Moreover, the same DNA sequence serves as a hormone response element for the activated (steroid-bound) forms of both mineralocorticoid and glucocorticoid receptors. An obvious question is:
How can aldosterone stimulate specific biological effects in this kind of system, particularly when blood concentrations of cortisol are something like 2000-fold higher than aldosterone?
A large part of the answer is that, in aldosterone-responsive cells, cortisol is effectively destroyed, allowing aldosterone to bind its receptor without competition. Target cells for aldosterone express the enzyme 11-beta-hydroxysteroid dehydrogenase, which has no effect on aldosterone, but converts cortisol to cortisone, which has only a very weak affinity for the mineralocorticoid receptor. In essence, this enzyme "protects" the cell from cortisol and allows aldosterone to act appropriately. Some tissues (e.g. hippocampus) express abundant mineralocorticoid receptors but not 11-beta HSD - they therefore do not show responses to aldosterone because aldosterone is not present in quantities sufficient to compete with cortisol.
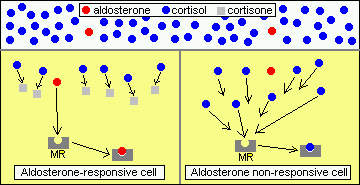
An interesting demonstration of this enzyme protection system is seen in chronic licorice intoxication.
The drug spironolactone inhibits the effects of aldosterone by competitively binding to the aldosterone receptor in target cells; this drug is most commonly used because of its effects on the kidney to promote water excretion.
Physiologic Effects of Mineralocorticoids
Mineralocorticoids play a critical role in regulating concentrations of minerals - particularly sodium and potassium - in extracellular fluids. As described above, loss of these hormones leads rapidly to life-threatening abnormalities in electrolyte and fluid balance.
The major target of aldosterone is the distal tubule of the kidney, where it stimulates exchange of sodium and potassium. Three primary physiologic effects of aldosterone result:
- Increased resorption of sodium: sodium loss in urine is decreased under aldosterone stimulation.
- Increased resorption of water, with consequent expansion of extracellular fluid volume. This is an osmotic effect directly related to increased resorption of sodium.
- Increased renal excretion of potassium.
Knowing these effects should quickly suggest the cellular mechanism of action this hormone. Aldosterone stimulates transcription of the gene encoding the sodium-potassium ATPase, leading to increased numbers of "sodium pumps" in the basolateral membranes of tubular epithelial cells. Aldosterone also stimulates expression of a sodium channel which facilitates uptake of sodium from the tubular lumen.
Aldosterone has effects on sweat glands, salivary glands and the colon which are essentially identical to those seen in the distal tubule of the kidney. The major net effect is again to conserve body sodium by stimulating its resorption or, in the case of the colon, absorption from the intestinal lumen. Conservation of water follows conservation of sodium.
Control of Aldosterone Secretion
Control over aldosterone secretion is truly multifactorial and tied into a spider web of other factors which regulate fluid and electrolyte composition and blood pressure. If the major effects of aldosterone are considered, it is rather easy to predict factors which stimulate or suppress aldosterone secretion.
The two most significant regulators of aldosterone secretion are:
- Concentration of potassium ions in extracellular fluid: Small increases in blood levels of potassium strongly stimulate aldosterone secretion.
- Angiotensin II: Activation of the renin-angiotensin system as a result of decreased renal blood flow (usually due to decreased vascular volume) results in release of angiotensin II, which stimulates aldosterone secretion.
Other factors which stimulate aldosterone secretion include adrenocorticotropic hormone (short-term stimulation only) and sodium deficiency. Factors which suppress aldosterone secretion include atrial naturetic hormone, high sodium concentration and potassium deficiency.
Disease States
A deficiency in aldosterone can occur by itself or, more commonly, in conjunction with a glucocorticoid deficiency, and is known as hypoadrenocorticism or Addison's disease. Without treatment by mineralocorticoid replacement therapy, a lack of aldosterone is lethal, due to electrolyte imbalances and resulting hypotension and cardiac failure.
Aldosterone excess is most commonly observed in two conditions: elevated plasma potassium (hyperkalemia) and low vascular volume. This should make sense considering that plasma potassium and angiotensin II are the major factors that regulate aldosterone secretion, as described above. Importantly, it is now recognized that roughly 1 in 10 cases of primary hypertension in humans is associated with hyperaldosteronism, due most commonly to aldosterone-secreting adrenal tumors or mutations in potassium channels.
Advanced and Supplemental Topics
Send comments to Richard.Bowen@colostate.edu

