VIVO Pathophysiology
Insulin-like Growth Factors (IGFs)
Insulin-like growth factors or IGFs are peptide hormones secreted from many different cells. Their designation as "insulin-like" originated from experiments in which treatment of serum with antibodies to insulin failed to eliminate all insulin activity; the remaining activity was ultimately ascribed to the IGFs. Due to their growth promoting activity, they were formerly called somatomedins.
IGFs, IGF Receptors and IGF-Binding Proteins
There are two principle IGFs referred to as IGF-1 and IGF-2. Each of these has a number of variant forms, resulting from use of alternative gene promoters and alternative splicing. Structurally, both IGFs resemble insulin in having two chains (A and B) connected by disulfide bonds. Human IGF-1 and IGF-2 are, respectively, 70 and 67 amino acids in length.
In addition to two IGFs, there are three receptors that bind IGFs with differing affinities:
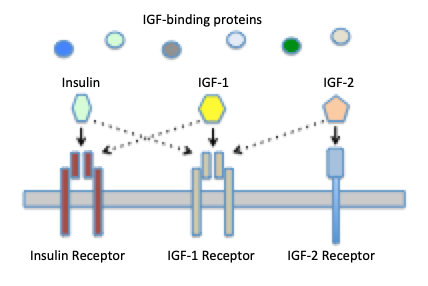
- IGF-1 receptor: binds both IGF-1 and IGF-2 with high affinity. This receptor has been identified in essentially all tissues except liver, and virtually all of the biological activities of the IGFs result from binding to the type 1 receptor. This receptor is a tyrosine kinase type of cell surface receptor very similar to that for insulin.
- IGF-2 receptor: binds IGF-2 with high affinity and IGF-1 with low affinity. It appears primarily to be involved in clearance and degradation of IGF-2 and is not involved in signal transduction. This is also the cation-independent mannose-6-phosphate receptor used for targeting mannosylated enzymes to lysosomes.
- Insulin receptor: binds IGF-I with roughly 100-fold lower affinity than insulin. High concentrations of IGF may stimulate insulin signalling through this receptor.
Interestingly, the IGF-1 and insulin receptors are very similar structurally. In fact, in cells that express both receptors, hybrid receptors appear to readily form, although the biological consequences of these hybrids is not clear.
A final important determinant of IGF activity is a family if IGF-binding proteins. IGFs circulate in blood complexed to these proteins, which not only extend the half-life of the hormones, but modulate their interaction with receptors. To date, at least six distinct IGF-binding proteins have been identified in humans and rats.
It should not come as a surprise that this endocrine system is highly complex, as it involves interactions among three related hormones (IGF-1, IGF-2, insulin), three cross-binding receptors, and a multitude of IGF binding proteins.
IGF-1: Secretion and Biological Activity
Roughly 75% of the IGF-1 circulating in blood is synthesized and secreted from hepatocytes in the liver in response to stimulation by growth hormone. Additionally, virtually all other tissues secrete IGF-1 that has autocrine or paracrine effects locally.
IGF-1 is the hormone largely responsible for the growth-promoting properties of growth hormone, and one of the major controllers of growth hormone secretion is the negative feedback effect from IGF-1. In addition to its clear role in promoting growth, a large number of other effects have been ascribed to IGF-1. These effects can be crudely lumped into categories of growth, cellular differentiation, cell proliferation, and cell survival. Obviously, these processes are highly complex and dependent on numerous factors other than IGFs. Overall, it is safe to state that there are few tissues or physiologic systems that are not affected to some extent by IGF-1.
IGF-2: Secretion and Biological Activity
Like IGF-1, IGF-2 is widely expressed in the body and secreted from the liver. IGF-2 is also secreted abundantly from the placenta. Both IGFs are clearly important in regulating growth, and the general observation is that IGF-2 is more important for prenatal (fetal) growth, while IGF-1 assumes a major role in growth after birth. Mice with targeted disruptions of either IGF gene show significant reductions in body size and birth, but those with disrupted a IGF-2 gene are much more severe.
Interestingly, the gene encoding IGF-2 is imprinted such that only the paternal allelle is expressed. Mutations affecting imprinting of the IGF-2 gene have been identified that result either in enhanced expression (Beckwith-Wiedemann syndrome), leading to fetal overgrowth, or deficient expression (Russell-Silver syndrome), which results in growth restriction. These genetic disorders clearly emphasize the importance of IGF-2 in prenatal growth.
Disease States
A dramatic but rare manifestion of IGF-1 deficiency in humans is referred to as Laron dwarfism. Children with this condition are close to normal size at birth, but experience dramatically slow grow during childhood, and without treatment, usually achieve a maximal height of roughly 4 feet. Laron syndrome is clinically indistinguishable from growth hormone deficiency, and can result from a number of genetic defects. Most commonly, affected patients have mutations in the growth hormone receptor gene. As would be expected, this receptor deficiency is associated with low serum levels of IGF-1 (lack of growth hormone stimulation of liver cells) and high serum levels of growth hormone (lack of negative feedback by IGF-1). Additional causes of Laron syndrome include defects in growth hormone signal transduction, defects in IGF-1 synthesis, and IGF-1 receptor deficiency. A similar syndrome is seen in African pygmies, who also have short stature due to congenital IGF-1 deficiency; it this case, there is a dramatic reduction in the expression of the growth hormone receptor gene and hence, the number of growth hormone receptors.

Consider dogs - Canis lupus familiaris. There is no other species that shows such dramatic variation in body size, ranging from Chihuahuas to Great Danes, and clearly the results of artificial selection. Studies have revealed that mean plasma concentrations of IGF-1 in standard poodles was approximately six times that of miniature poodles, and even lower in toy poodles. Subsequent research demonstrated that small breed dogs overwhelmingly harbor variants of genes for both IGF-1 and the IGF-1 receptor that are only rarely found in large breed dogs.
As indicated above, a large fraction of circulating IGF-1 comes from heptatocytes in the liver, and liver disease - particularly chronic cirrohis - is associated with significant reductions in IGF-1.
Finally, it should be noted that we all become relatively deficient in IGF-1 with age. Concentrations of this hormone peak during late teenage and early adult ages, then steadily decline as individuals age.
References and Reviews
- Eigenmann JE, Patterson DF, Froesch ER. Body size parallels insulin-like growth factor I levels but not growth hormone secretory capacity. Acta Endocrinol (Copenh) 1984;106:448-53.
- Plassais J. et al. Natural and human-driven selection of a single non-coding body size variant in ancient and modern canids. Current Biology 2021; 32:1-9, https://doi.org/10.1016/j.cub.2021.12.036
- Puche JE1, Castilla-Cortázar I. Human conditions of insulin-like growth factor-I (IGF-I) deficiency. J Transl Med. 2012; 10:224.
- Le Roith D. Insulin-like growth factors. N Engl J Med 1997; 336:633-640.
- Hoopes BC, Rimbault M, Liebers D, Ostrander EA, Sutter NB. The insulin-like growth factor 1 receptor (IGF1R) contributes to reduced size in dogs. Mamm Genome 2012; 23:780-90.
Updated February 2022. Send comments to Richard.Bowen@colostate.edu
