VIVO Pathophysiology
Exocrine Secretions of the Pancreas
Pancreatic juice is composed of two secretory products critical to proper digestion: digestive enzymes and bicarbonate. The enzymes are synthesized and secreted from the exocrine acinar cells, whereas bicarbonate is secreted from the epithelial cells lining small pancreatic ducts.
Digestive Enzymes
The pancreas secretes a magnificent battery of enzymes that collectively have the capacity to reduce virtually all digestible macromolecules into forms that are capable of, or nearly capable of being absorbed. Three major groups of enzymes are critical to efficient digestion:
1. Proteases
Digestion of proteins is initiated by pepsin in the stomach, but the bulk of protein digestion is due to the pancreatic proteases. Several proteases are synthesized in the pancreas and secreted into the lumen of the small intestine. The two major pancreatic proteases are trypsin and chymotrypsin, which are synthesized and packaged into secretory vesicles as the inactive proenzymes trypsinogen and chymotrypsinogen.
As you might anticipate, proteases are rather dangerous enzymes to have in cells, and packaging of an inactive precursor is a way for the cells to safely handle these enzymes. The secretory vesicles also contain a trypsin inhibitor which serves as an additional safeguard should some of the trypsinogen be activated to trypsin; following exocytosis this inhibitor is diluted out and becomes ineffective - the pin is out of the grenade.
Once trypsinogen and chymotrypsinogen are released into the lumen of the small intestine, they must be converted into their active forms in order to digest proteins. Trypsinogen is activated by the enzyme enterokinase, which is embedded in the intestinal mucosa.
Once trypsin is formed it activates chymotrypsinogen, as well as additional molecules of trypsinogen. The net result is a rather explosive appearance of active protease once the pancreatic secretions reach the small intestine.
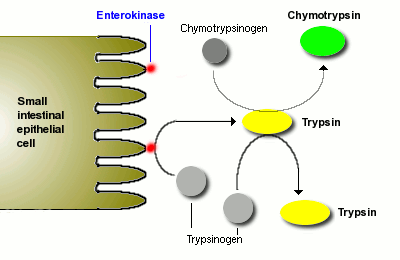
Trypsin and chymotrypsin digest proteins into peptides and peptides into smaller peptides, but they cannot digest proteins and peptides to single amino acids. Some of the other proteases from the pancreas, for instance carboxypeptidase, have that ability, but the final digestion of peptides into amino acids is largely the effect of peptidases on the surface of small intestinal epithelial cells. More on this later.
2. Pancreatic Lipase
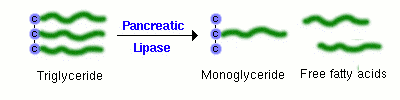
A major component of dietary fat is triglyceride, or neutral lipid. A triglyceride molecule cannot be directly absorbed across the intestinal mucosa. Rather, it must first be digested into a 2-monoglyceride and two free fatty acids. The enzyme that performs this hydrolysis is pancreatic lipase, which is delivered into the lumen of the gut as a constituent of pancreatic juice.
Sufficient quantities of bile salts must also be present in the lumen of the intestine in order for lipase to efficiently digest dietary triglyceride and for the resulting fatty acids and monoglyceride to be absorbed. This means that normal digestion and absorption of dietary fat is critically dependent on secretions from both the pancreas and liver.
Pancreatic lipase has recently been in the limelight as a target for management of obesity. The drug orlistat (Xenical) is a pancreatic lipase inhibitor that interferes with digestion of triglyceride and thereby reduces absorption of dietary fat. Clinical trials support the contention that inhibiting lipase can lead to significant reductions in body weight in some patients.
3. Amylase
The major dietary carbohydrate for many species is starch, a storage form of glucose in plants. Amylase (technically alpha-amylase) is the enzyme that hydrolyses starch to maltose (a glucose-glucose disaccharide), as well as the trisaccharide maltotriose and small branchpoints fragments called limit dextrins. The major source of amylase in all species is pancreatic secretions, although amylase is also present in saliva of some animals, including humans.
Other Pancreatic Enzymes
In addition to the proteases, lipase and amylase, the pancreas produces a host of other digestive enzymes, including ribonuclease, deoxyribonuclease, gelatinase and elastase.
Bicarbonate and Water
Epithelial cells in pancreatic ducts are the source of the bicarbonate and water secreted by the pancreas. Bicarbonate is a base and critical to neutralizing the acid coming into the small intestine from the stomach. The mechanism underlying bicarbonate secretion is essentially the same as for acid secretion by parietal cells in the stomach and is dependent on the enzyme carbonic anhydrase. In pancreatic duct cells, the bicarbonate is secreted into the lumen of the duct and hence into pancreatic juice.
Send comments to Richard.Bowen@colostate.edu
A Ukrainian translation of this page by Olena Chervona is available at Ukrainian translation

